On MND Global Awareness Day, Professor Jackie de Belleroche looks at how increased awareness and collaboration, alongside advances in genetic and molecular research, offers hope for the future.
Without doubt, the last few years have seen phenomenal developments not only in research, but also in public awareness of just how devastating a condition motor neurone disease (MND) is. There is hardly anyone who has not heard of MND thanks to Professor Stephen Hawking, well-known to the public as an outstanding scientist who was diagnosed with the condition at the age of 21. The ALS “ice bucket challenge” certainly caught the imagination of the international community too, as well as raising £88m in a single month for MND research across the world (it’s important to note here that MND is usually referred to as amyotrophic lateral sclerosis [ALS] outside the UK).
MND is the most common neuromuscular disorder in adults. In essence, it targets the motor system and affects our ability to use our arms and legs, and to speak and swallow. Above all, it generally has a very rapid progression compared to other common neurological disorders, such as Parkinson’s disease and Alzheimer’s disease.

Making advances through the power of international collaboration
Over the last few years, the wider scientific community has obtained significant insights into the mechanisms that underlie MND, most notably the identification of the gene mutations that cause the familial form of the disease (meaning that it is passed from generation to generation). Although only accounting for 10% of MND cases, the number of disease-causing genes has steadily increased since 2008, rising to over 30 genes at present; these mutations are responsible for two thirds of familial MND cases worldwide.
In discovering these gene mutations, we have been able to pinpoint at the molecular level what goes wrong in MND. In summary, these mutations cause abnormalities in the normal production of cellular proteins (essential molecules that are required for the structure, function, and regulation of the body’s tissues and organs), and the disposal of damaged protein, which ultimately leads to the death of motor neurones – the nerve cells that control our ability to move.
Our lab was the first in the UK to start recruiting families with familial MND, initially with a grant from the Motor Neurone Disease Association and later with support from the American ALS Association. Over time, we were gradually able to build up a cohort of over 200 families, which crucially allowed us to identify mutations that were transmitted with the disease. Although the techniques for doing this have developed beyond recognition – especially in terms of genome sequencing – one of the most important aspects contributing to the speed of research progress has been the value of international collaboration. Great advances have occurred through the pooling of resources, and some of the most important findings have come from large scale collaborations, for example, the familial ALS (FALS) international sequencing consortium.
A new phase of research
We have now entered a new phase of research, which is directed at probing the effects of these MND-related gene mutations. To do this, we’re using these mutations to create a model of the disease process at the cellular level. Having such a model system has allowed us to test whether a mutation in a specific molecular target is responsible for setting off the chain of events that causes the disease. One particular example is an MND-associated mutation in an enzyme known as D-amino acid oxidase (DAO). DAO is not necessarily a household name, but it plays a vital role by targeting the removal of a specific kind of amino acid known as D-serine, essential for some types of memory, but toxic if it builds up at the synapse.
In our investigation, we noted that a specific DNA mutation prevents it from working as it typically would, removing build-up of D-serine at the synapse. To investigate whether the build-up of D-serine caused by loss of the activity from this important enzyme could trigger the disease process, we used a drug to block the action of D-serine at the synapse. We found that we could significantly weaken the toxic effects of D-serine and subsequent death of motor neurones, thereby instigating the progression of MND at a cellular level. This is only the beginning, but it suggests that this activity might trigger the disease, and could therefore be used as a starting point to create new therapies. Furthermore, other research has shown that levels of D-serine – the amino acid targeted by DAO – are elevated in sporadic cases of MND, suggesting that this may be a common mechanism underlying both sporadic and familial forms of the disease.
Exploring molecular pathways
Another approach that we are using is to explore the molecular pathways revealed by our mutation analysis in the non-familial forms of MND, which make up the majority cases (90%). In other words, we’re looking at the series of interactions between molecules that cause the cells to change, ultimately leading to the development of MND. Many of these pathways regulate protein folding (the process by which a protein structure assumes its functional shape), facilitate protein movement within the cell, or cause protein degradation. Using a combined gene expression and bioinformatics approach, we have quantified the key players in these pathways, which we again hope can be used as potential therapeutic targets in the future.
Find out more about MND Global Awareness Day, which takes place annually on June 21.
Jackie de Belleroche is a Professor in the Department of Medicine and Course Director for Imperial’s BSc Course in Neuroscience and Mental Health. Her research focuses on Amyotrophic Lateral Sclerosis (ALS), and the main aim of her work has been to identify disease genes associated with the dominantly inherited familial form of ALS (FALS). Professor de Belleroche has been a member of the Motor Neurone Disease Association’s Research Advisory Panel (2009-2013), and several international scientific committees for research on amyotrophic lateral sclerosis.
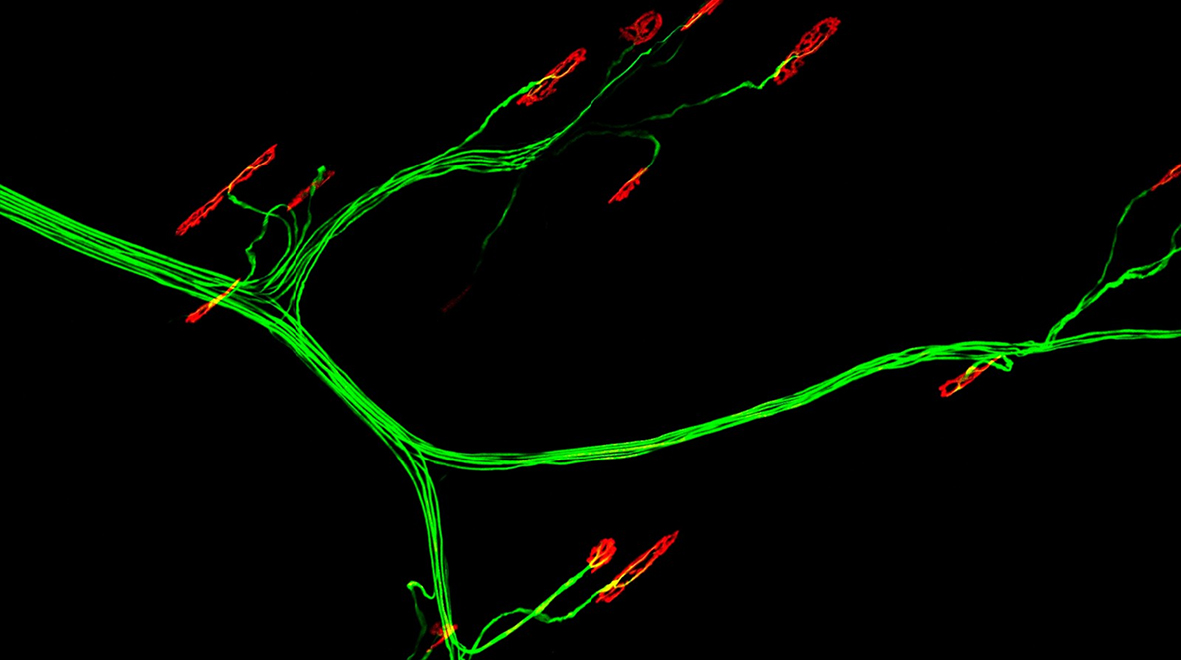
Image description: Motor nerves (green) found in the spinal cord project out long, thin axons to the periphery, where they contact muscles (black background), forming specialised connections or synapses called neuromuscular junctions (NMJs, red)
Credit: James N. Sleigh / Wellcome Images
Following the launch of the Faculty of Medicine’s reorganised academic structure on 1 August 2019, this post was recategorised to Department of Brain Sciences.
Came across you article while looking into L-serine and ALS.
Was wondering why no mention of L-serine for ALS?
If you think it may be an useful path for ALS / Parkisons/ Alzheimers as some web sites are claiming?
https://n.neurology.org/content/88/16_Supplement/P3.128